Stevens research finds that cyclopropanation reactions sometimes take the scenic route
Sometimes, serendipity—or just plain luck—still plays a pivotal role in scientific discovery. Recently, a team of chemists were experimenting with using a biocatalytic process to trigger a cyclopropanation reaction, which yields intricate molecular structures used in various drugs and other natural processes, when something unusual happened: a beaker of liquid that should have shownmuddy red turned bright green instead.
The team—which grew to include researchers from the Stevens Institute of Technology alongside colleagues from Oxford, Cornell, Rochester, and the University of Texas—published their explanation of the weird result in Nature Communications. Their key finding: even when chemical reactions are apparently well-understood, sometimes nature takes the scenic route.
“It turns out that you can’t assume there’s just one chemical pathway at work,” explains Dr. Yong Zhang, a professor in the Department of Chemistry and Chemical Biology at Stevens. “When we looked more closely, we found that some unexpected and entirely new chemistry was taking place.”
The reaction in question used engineered hemoproteins—like the hemoglobin and myoglobin that store and transport oxygen in our blood and muscles—as the catalyst in a carbene transfer reaction. The goal: to use carbene, an extremely reactive carbon-based molecule, to generate a cyclopropane ring, which could then be used to generate a wide range of useful compounds including a number of important antibiotics.
Using hemoproteins to trigger that reaction is attractive because they contain the abundant Fe as the metal center and are nontoxic, easy to fine-tune, and viable at room temperature. “That’s exactly what you need in order to manufacture drugs safely and cheaply,” explains Dr. Zhang. “But if it turns out you’re also producing unexpected molecules of other kinds, it could create safety issues or compromise the efficiency of the reaction.”
To understand the unexpected chemical reaction, the researchers used a range of cutting-edge techniques to explore the chemistry at work, including advanced computational methods led by Dr. Zhang. Other research units used techniques such as Mössbauer spectroscopy and X-ray crystallography to characterize molecules at the atomic scale and accurately map the precise structures involved in combination with calculations.
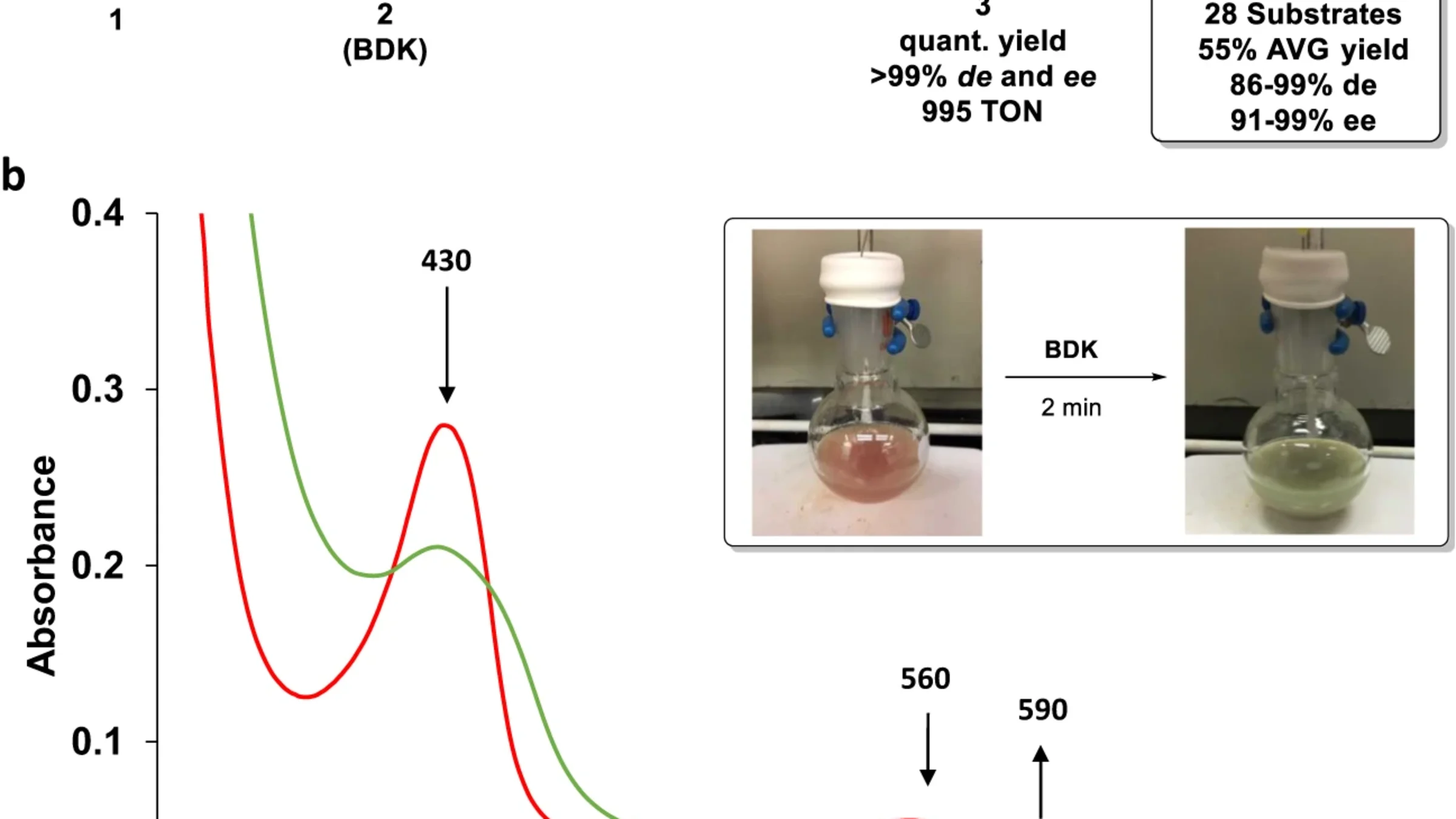
Collectively, the team’s work revealed that while in many cases cyclopropane rings were being formed via the intended hemoprotein-mediated carbene reaction, a number of other complex and unanticipated reactions were also taking place. In some cases, computational methods revealed the possibility of rare side-reactions that required more energy than is typically available in a room-temperature reaction. In others, computational and observational methods identified multiple competing pathways out of which cyclopropanation could occur.
Notably, one alternate pathway led to the formation of a myoglobin variant complex called Mb-cIII —the strange green compound the team had initially found in their beaker. Further research showed that the Mb-cIII complex naturally regenerated the hemoprotein catalysts used in the original reaction, allowing the intended cyclopropanation reaction to continue.
“What we found wasn’t a dead end, but rather a detour,” Dr. Zhang said. “It’s a sign that these important reactions aren’t simple and linear, but rather a complex network of forking chemical pathways.”
The team’s ability to untangle those pathways and describe the full complexity of the cyclopropanation reaction should streamline important future research on carbene-based reactions and biocatalysts, Dr. Zhang says. Crucially, it also shows the value of using both computational and experimental methods to explore chemical reactions, and to ensure that all possible side-reactions are considered when exploring new chemistry.
“As scientists, we need to remain open-minded,” Dr. Zhang says. “If you assume you already understand exactly how these processes work, then you limit your opportunity to discover new things.”
About Stevens Institute of Technology
Stevens Institute of Technology is a premier, private research university situated in Hoboken, New Jersey. Since our founding in 1870, technological innovation has been the hallmark of Stevens’ education and research. Within the university’s three schools and one college, 8,000 undergraduate and graduate students collaborate closely with faculty in an interdisciplinary, student-centric, entrepreneurial environment. Academic and research programs spanning business, computing, engineering, the arts and other disciplines actively advance the frontiers of science and leverage technology to confront our most pressing global challenges. The university continues to be consistently ranked among the nation’s leaders in career services, post-graduation salaries of alumni and return on tuition investment.
Stevens Media Contact
Kara Panzer
Interim Director of Public Relations
Division of University Relations
845-475-4594
[email protected]